Presentation on cardiac system of human body
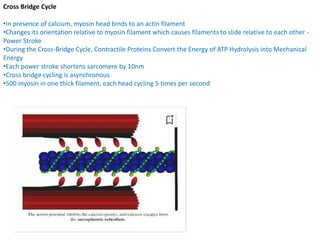
- 1. Cross Bridge Cycle •In presence of calcium, myosin head binds to an actin filament •Changes its orientation relative to myosin filament which causes filaments to slide relative to each other - Power Stroke •During the Cross-Bridge Cycle, Contractile Proteins Convert the Energy of ATP Hydrolysis into Mechanical Energy •Each power stroke shortens sarcomere by 10nm •Cross bridge cycling is asynchronous •500 myosin in one thick filament, each head cycling 5 times per second
- 2. Cross Bridge Cycle Occurs in 5 steps:- 1.Cross - Bridge formation • cocked myosin head (perpendicular or at a 90-degree angle to the thick and thin filaments) binds to actin filament • Cocked head has the stored energy derived from the cleaved ATP Cross Bridge Cycle 2.Release of Pi from the myosin • Dissociation of Pi from the myosin head triggers power stroke • Conformational change - myosin head bends approximately 450 about the hinge • Pulls the actin filament about 11 nm toward the tail of the myosin molecule • Generating force and motion.
- 3. Cross Bridge Cycle 3.ADP release – • Dissociation of ADP from myosin • Myosin head remains in the same position (45° angle with respect to the thick and thin filaments) 4.ATP binding – • ATP binding to the head of the myosin heavy chain (MHC) reduces the affinity of myosin for actin • Myosin head releases actin filament • Cross Bridge Cycle 5.ATP hydrolysis • Breakdown of ATP to ADP and inorganic phosphate (Pi) occurs on myosin head • Products of hydrolysis are retained on the myosin • As a result of hydrolysis, the myosin head pivots around the hinge into a "cocked" position (perpendicular or at a 900 angle to the thick and thin filaments) • Rotation causes the tip of the myosin to move about 11 nm along the actin filament so that it now lines up with a new actin monomer two monomers further along the actin filament
- 4. Cross Bridge Cycle • Cycle repeats as long as Ca, is elevated and sufficient ATP is there • Muscle cells do not regulate cross-bridge cycling by modifying [ATP]i • Instead, skeletal muscle and cardiac muscle control this cycle by preventing cross- bridge formation until the tropomyosin moves out of the way in response to an increase in [Ca']i
- 8. Steps in Relaxation • Cell may extrude Ca,- using either an Na-Ca exchanger (NCX) or a Ca' pump(PMCA) • However, would eventually deplete the cell of Ca, and is thus a minor mechanism for Ca, removal from the cytoplasm • Instead, Ca, re-uptake into the SR is the most important mechanism by which the cell returns [Ca']i to resting levels • Ca' re-uptake by the SR is mediated by a SERCA(s arcoplasmic or e ndoplasmic r eticulum C a2+8 TPase )-type Ca' pump
- 9. Steps in Relaxation • SR Ca2+-pump activity is inhibited by high [Ca2*] within the SR lumen • Inhibition of SR • Ca2+-pump activity is delayed by • Ca?+-binding proteins within the SR lumen = Buffer the Ca?* increase in the SR during Ca* re-uptake and thus markedly increase the Ca2* capacity of SR = Proteins have a tremendous capacity to bind Ca?* with up to 50 binding sites per protein molecule • Principal Ca2* binding protein in skeletal muscle, calsequestrin • Also present in cardiac and some smooth muscle • Calreticulin - Ca?*-binding protein found in particularly high concentrations within the SR of smooth muscle
- 10. Steps in Relaxation • When Ca*+ conc. outside has lowered, interaction of actin & myosin ceases & muscle relaxes • ATP required for both contraction &relaxation • Pump concentrates Cat+ about 10,000fold • Normal/Resting Ca*+ conc. (less than 10-7 moles of Cat** ) rises to 10°° • Total duration of Ca*+ ions stay in fluid is 1/30th of sec • Contracture • Ca?* movement inhibited • Relaxation fails to occur • Cross bridges don’t break • Sustained contraction despite no action potential
- 11. Role of ATP • Provides energy for power stroke of myosin head • Brings about a dissociation of myosin head from actin filament • Brings about muscle relaxation by pumping Ca2*+ back into sarcoplasmic reticulum Rigor Mortis • Muscles of body become very stiff and rigid shortly after death • Due to loss of ATP in the muscle cell • In absence of ATP, the myosin cross bridges with actin is not broken, so, no relaxation occurs • 15-25 hrs later, muscle proteins deteriorate and rigor disappears
- 13. EC Coupling: Drugs • Blocking release of Ca++ from SR keeps muscle relaxed, even in the presence of action potential eg Ryanodine receptor blocker like Protamine sulphate • Caffeine cause release of Ca++ produces contraction without action potential • Drugs which increase the release of Ca++ from sarcoplasmic reticulum. eg. Digitalis increase the force of cardiac muscle contraction
- 14. Malignant Hyperthermia • Channelopathy of calcium release channel in muscle (Ryanodine receptors • constant leak of SR Ca2+ through ryanodine receptor • triggered by halogenated anesthetics (isoflurane, halothane) or severe exercise familial tendency - can be tested for by muscle biopsy Symptoms • Normal muscle function under normal conditions • increased body temperature -more heat produced = skeletal muscle rigidity • lactic acidosis (hypermetabolism)
- 15. Troponin Each troponin C molecule in skeletal muscle has • 2 high-affinity Ca2* -binding sites • 2 low-affinity Ca?*-binding sites Binding of Ca?* to low-affinity sites induces a conformational change in the troponin complex that has two effects • Troponin I moves away from the actin/tropomyosin filament, thereby permitting the tropomyosin molecule to move • Troponin T pushes tropomyosin away from the myosin- binding site on the actin and into the actin groove • With the steric hindrance removed, the myosin head is able to interact with actin and engage in cross-bridge cycling
- 16. CARDIAC MUSCLES • EXCITATION-CONTRACTION COUPLING MECHANISM GAP JUNCTIONS • Proteinaceous tubes that connect cells • Permit ions to flow from one cell to another without having to pass from plasma membrane of the cell • Dissolved substances such as glucose, ions can pass through gap junctions
- 17. SYNCYTIUM • Action potential can pass from one direction to • Interconnections ■ Two kinds of syncytiums ■ Atrial syncytium • Constitute the walls of 2 atria ■ Ventricular syncytium ■ Constitute the walls of 2 ventricles BARRIERS TO CONDACTION • Separation of atria from ventricles by means of fibrous band tissue • High electrical resistance • Propagation of myocardial action potential from atria to ventricles • By specialized conductive system called AV node or AV bundle
- 18. IMPORTANT SEQUENCE TWO SYNCYTIUM STRUCTURES • The separate structures allow the atria to contract in time slightly before the ventricles • A condition central to cardiac pumping
- 19. • IMPORTANT SEQUENCE 0 TWO SYNCYTIUM STRUCTURES • The separate structures allow the atria to contract in time slightly before the ventricles • A condition central to cardiac pumping effectiveness
- 20. • ACTION POTENTIAL IN CARDIAC MUSCLES • Resting membrane potential -85mv to -90mv for normal cardiac muscles -90 to -100mv for specialized fibers e.g bundle of his, AV nodal fibers Magnitude for ventricular muscles about 105mv i.e going from about -85mv to +20mv IONS AND MYOCARDIAL ACTION POTENTIAL • Abrupt change in membrane potential • Corresponds to initial spike • Depolarization duration=about 0.2 s for atrial muscle and 0.3 sec for ventricular muscles • Plateau phase • Extends the duration of action potential significantly 3-15 times more as compared to skeletal muscles
- 21. IONS AND MYOCARDIAL ACTION POTENTIAL • Sudden opening of fast sodium channels • Allow to enter sodium ions from extracellular fluid • Fast channels • Duration of opening • Few thousands of sec • Abrupt closure SLOW RESPONSE DUE TO ACTIVATION OF SLOW CALCIUM CHANNELS • Fast sodium channels • Slow calcium channels • Also called calcium-sodium channels • Slower to open • Remain open for several tenths of sec Prolonged period of contraction • Formation of plateau
- 22. RECOVERY OF RESTING MEMBRANE POTENTIAL • Inactivation of sodium-calcium channels • Membrane permeability of potassium increases • Potassium efflux results in repolarization increases MYOCARDIAL ACTION
- 23. ACTION POTENTIAL SPREAD • Over the surface • To interior of fiber along transverse tubules EXCITATION-CONTRACTION COUPLING MECHANISM • The mechanism by which the action potential causes the myofibrils of muscles to contract • Sarcoplasmic reticulum as a source of calcium ions in skeletal muscles • Diffusion of large quantity of extra calcium ions from T-tubules • No contraction of cardiac muscles
- 24. EXCITATION-CONTRACTION COUPLING MECHANISM • Less developed sarcoplasmic reticulum • T-tubules have diameter 5 times great as compared to skeletal muscles • 25 times great volume • A large quantity of mucopolysaccharides • Electronegativity charged • Binding with abundant source of calcium ions • Concentration of calcium ions in extracellular fluid • Opening of T-tubules pass directly through cell membrane into extracellular spaces
- 25. EXCITATION-CONTRACTION COUPLING MECHANISM • Permission of percolation of calcium ions from extracellular fluid • Dependence of contraction on calcium ions from extracellular fluid • Comparison to skeletal muscles • Cessation of contraction • Pump system • T-tubules extracellular fluid OVERALL VIEW OF CONTRACTION OF SKELETAL AND CARDIAC MUSCLES • SKELETAL MUSCLES • Highly developed sarcoplasmic reticulum • Less developed T-system • No electronegativity Extracellular fluid is not efficien • CARDIAC MUSCLES • Less developed • sarcoplasmic reticulum Highly developed T- system • Electronegativity • Extracellular fluid is efficient and major source of calcium ions