A Practical Guide for Making Multidisciplinary Decisions About Neoadjuvant and/or Adjuvant Immunotherapy in Resectable NSCLC
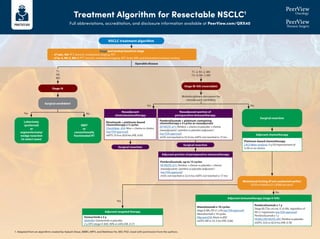
- 1. Treatment Algorithm for Resectable NSCLC1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/QRX40 Stage and workup based on stage • cT1abc, N0: PFT, bronch, mediastinal staging, PET • cT2a-4, N0-3, M0-1: PFT, bronch, mediastinal staging, PET, brain MRI, and biomarker/mutation testing Surgical candidate? Lobectomy (preferred) or segmentectomy/ wedge resection (in select cases) SBRT or conventionally fractionated RT Surgical resection Mutational testing (if not conducted earlier) EGFR ex19del/ex21 L858R present? Surgical resection T1 N0 M0 Operable disease Yes Yes Yes No No No Multidisciplinary discussion for neoadjuvant candidacy T1–2, N1–2, M0 T3–4, N0–1, M0 Neoadjuvant chemoimmunotherapy Neoadjuvant portion of perioperative immunotherapy Nivolumab + platinum-based chemotherapy x 3 cycles CheckMate -816: Nivo + chemo vs chemo (see FDA approval) mEFS: 31.6 vs 20.8 mo (HR, 0.63) Pembrolizumab + platinum-containing chemotherapy x 4 cycles as neoadjuvant KEYNOTE-671: Pembro + chemo vs placebo + chemo (neoadjuvant) / pembro vs placebo (adjuvant ) (see FDA approval) mOS: not reached vs 52.4 mo; mEFS: not reached vs 17 mo Adjuvant chemotherapy Platinum-based chemotherapy LACE Meta-analysis: 5-y OS improvement of 5.4% vs no chemo Adjuvant immunotherapy (stage II-IIIA) Atezolizumab x 16 cycles StageII-IIIA,PD-L1≥1%(seeFDAapproval) Atezolizumab x 16 cycles IMpower010: Atezo vs BSC mDFS: NR vs 35.3 mo (HR, 0.66) Pembrolizumab x 1 y Stage IB (T2a ≥4 cm), II, or IIIA, regardless of PD-L1 expression (see FDA approval) Pembrolizumab x 1 y PEARLS/KEYNOTE-091: Pembro vs placebo mDFS: 53.6 vs 42.0 mo (HR, 0.76) Adjuvant targeted therapy Osimertinib x 3 y ADAURA: Osimertinib vs placebo 2-y DFS (stage II-IIIA): 90% vs 44% (HR, 0.17) NSCLC treatment algorithm Stage IB-IIIA (resectable) Stage IA Surgical resection Adjuvant portion of perioperative immunotherapy Pembrolizumab, up to 13 cycles KEYNOTE-671: Pembro + chemo vs placebo + chemo (neoadjuvant) / pembro vs placebo (adjuvant ) (see FDA approval) mOS: not reached vs 52.4 mo; mEFS: not reached vs 17 mo 1. Adapted from an algorithm created by Aakash Desai, MBBS, MPH, and Matthew Ho, MD, PhD. Used with permission from the authors.
- 2. Immune-Related Adverse Events of Cancer Immunotherapies Become Aware and Stay Vigilant Full abbreviations, accreditation, and disclosure information available at PeerView.com/QRX40 What Are irAEs?1 • Immune checkpoint inhibitors are associated with important clinical benefits, but general immunologic enhancement can also lead to a unique spectrum of immune-related adverse events • Any organ system can be affected, but more commonly occurring are pulmonary (pneumonitis), dermatologic (rash, pruritus, blisters, ulcers, vitiligo), gastrointestinal (diarrhea, enterocolitis, transaminitis, hepatitis, pancreatitis), and endocrine (thyroiditis, hypophysitis, adrenal insufficiency) irAEs Endocrine Hyper- or hypothyroidism Hypophysitis Adrenal insufficiency Diabetes Hepatic Hepatitis Renal Nephritis Dermatologic Rash Pruritus Psoriasis Vitiligo DRESS Stevens-Johnson Hematologic Hemolytic anemia Thrombocytopenia Neutropenia Hemophilia Ocular Uveitis Conjunctivitis Scleritis, episcleritis Blepharitis Retinitis Respiratory Pneumonitis Pleuritis Sarcoid-like granulomatosis Cardiovascular Myocarditis Pericarditis Vasculitis Gastrointestinal Colitis Ileitis Pancreatitis Gastritis Neurologic Neuropathy Guillain Barŕe Myelopathy Encephalitis Myasthenia Musculoskeletal Arthritis Dermatomyositis 01 Prevention 02 Anticipation 03 Detection 04 Treatment 05 Monitoring 01 02 03 04 05
- 3. Immune-Related Adverse Events of Cancer Immunotherapies Become Aware and Stay Vigilant Full abbreviations, accreditation, and disclosure information available at PeerView.com/QRX40 General Recommendations for Treating irAEs2-5 Increasing intensity of treatment required Grade 2 Grade 1 Grade 3 Grade 4 Moderate Mild Severe Very severe Symptomatic supportive therapy Stop treatment Oral steroids Intravenous steroids. ------------ • Referral to specialist • Strong immune suppressive treatment Increasing grade of irAE intravenous steroids Steroids (PO/IV): 1-2 mg/kg/d prednisone or equivalent, slowly taper over 4-6 weeks For some AEs, treatment can be restarted after resolution (eg, rash); generally, ICI can be continued with endocrinopathies once managed Managed in outpatient/ community setting Generally requires hospital admission
- 4. Immune-Related Adverse Events of Cancer Immunotherapies Become Aware and Stay Vigilant Full abbreviations, accreditation, and disclosure information available at PeerView.com/QRX40 Hold immunotherapy and reassess in 1-2 weeks Pulse oximetry rest and ambulation Consider chest imaging with CT (with contrast preferred) Repeat in 3-4 weeks Moderate (grade 2): 25%-50% lung involved Severe (grade 3-4) Grade 3: all lobes of lung or 50% of lung parenchyma; limited ADLs, oxygen requirement Grade 4: life threatening Hold immunotherapy Infectious workup (nasal swab, sputum, blood) Consider bronchoscopy and BAL Chest imaging with CT contrast Repeat in 3-4 weeks Consider empiric antibiotics Refractory: methylprednisolone 1-2 mg/m2 /day; if no response in 3-4 days, treat as grade 3 Permanently discontinue immunotherapy and move to inpatient care Infectious workup (nasal swab, sputum, blood) Pulmonary and infectious disease consultation Bronchoscopy with BAL Empiric antibiotics Methylprednisolone 1-2 mg/m2 /day; when grade 1, taper over 6 weeks Refractory: infliximab, mycophenolate, or IVIG How Should Pulmonary irAEs Be Diagnosed and Managed?2,6 Pneumonitis: focal or diffuse inflammation of the lung parenchyma (typically identified on CT imaging) Diagnostic workup: CXR, CT, pulse oximetry; for grade ≥2, may include infectious workup Mild (grade 1): 25% lung involved Additional considerations • GI and pneumocystis prophylaxis may be offered to patients on prolonged steroid use (12 weeks) • Consider calcium and vitamin D supplementation with prolonged steroid use • Bronchoscopy and biopsy; if clinical picture is consistent with pneumonitis, no need for biopsy Supportive care: smoking cessation and vaccinations (influenza, pneumococcal)
- 5. Immune-Related Adverse Events of Cancer Immunotherapies Become Aware and Stay Vigilant Full abbreviations, accreditation, and disclosure information available at PeerView.com/QRX40 1. Champiat S et al. Ann Oncol. 2016;27:559-574. 2. Brahmer JR et al. J Clin Oncol. 2018;36:1714-1786. 3. https://www.esmo.org/content/download/124130/2352601/1/ESMO-Patient-Guide-on-Immunotherapy-Side-Effects.pdf. 4. NCCN Clinical Practice Guidelines in Oncology. Management of Immunotherapy-Related Toxicities. Version 3.2023. https://www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf. 5. Puzanov I et al. J Immunother Cancer. 2017;5:95. 6. Provided courtesy of Marianne Davies, DNP, ACNP, AOCNP, FAAN, 2021; adapted from AIM with Immunotherapy, NCCN, and CTCAE. 7. https://ascopubs.org/doi/full/10.1200/JCO.21.01440. 8. https://www.sitcancer.org/research/cancer-immunotherapy-guidelines/irae/immune-checkpoint-inhibitor-related-adverse-events. Additional Guideline Recommendations for Treating irAEs3,4,7,8
- 6. Overview of Key Phase 3 Clinical Trials Investigating Immunotherapies in Resectable NSCLC1,2 Full abbreviations, accreditation, and disclosure information available at PeerView.com/QRX40 1. https://clinicaltrials.gov. 2. https://www.fda.gov/drugs/resources-information-approved-drugs/oncology-cancer-hematologic-malignancies-approval-notifications. SURGERY SURGERY Neoadjuvant Immunotherapy (Approved) Study Neoadjuvant Regimen Adjuvant Regimen Adjuvant Immunotherapy (Approved) AEGEAN Durvalumab + chemo x 4 cycles Durvalumab ~1 year CheckMate -77T Nivolumab + chemo x 4 cycles Nivolumab ~1 year KEYNOTE-671 Pembrolizumab + chemo x 4 cycles Pembrolizumab ~1 year CheckMate -816 IMpower010 KEYNOTE-091 Nivolumab + chemo x 3 cycles Chemo atezolizumab ~1 y (PD-L1 ≥1%) Chemo (optional) pembrolizumab ~1 year FDA approved FDA approved FDA approved SURGERY
