medicinal chemistry Prodrugs.pptx final.pptx
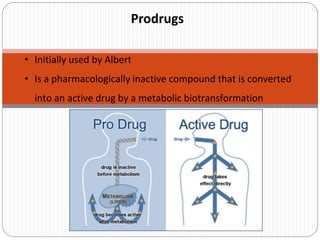
- 1. Prodrugs • Initially used by Albert • Is a pharmacologically inactive compound that is converted into an active drug by a metabolic biotransformation
- 2. • Can be enzymatic/non-enzymatic • Non-enzymatic such as hydrolysis- compounds may cause stability problems • Conversion can occur before ADME or at specific site in the body • Soft drug- pharmacologically active and uses metabolism for promotion of excretion
- 3. Why prodrug Lead modification approach used to correct a flaw in drug candidate •Aqueous solubility •Absorption and distribution •Site specificity •Instability •Prolonged release •Toxicity •Poor patient acceptability •Formulation problems
- 4. Types of Prodrugs • A) Carrier linked prodrugs and B) Bioprecursors A) Carrier linked prodrugs- active drug linked to a carrier group • Carrier group- should be labile, non-toxic, biologically inactive Further divided to bipartate, tripartate and mutual prodrugs 1. Bipartate- prodrug with carrier 2. Tripartate- carrier + linker + prodrug 3. Mutual prodrug- synergistic drugs connected to each other
- 5. Increase solubility Lipophilic isopropyl ester hydrolysed by corneal esterases
- 6. Ampicillin + CH2 (linker) + Pivalic acid carrier = Pivampicillin increase lipophilicity high bioavailability
- 7. Ampiciliin + Sulbactum (β lactamase inhibitor) = sultabactum (increase pharmacokinetics)
- 8. Sulfoxide converted to thioether GIT irritation Sulindac (Pro drug) converted to thioether by lever enzyme Low GIT irritation
- 9. Types of prodrugs • B) Bioprecursors- compound metabolized by molecular modification into new compound which can be drug • No resemblance to desired functional group • Drastic structural change is required to unmask desired group • Oxidation is common metabolic biotransformation
- 10. Carrier linked prodrugs • An ideal drug carrier must • (1) protect the drug until it is at the site of action; • (2) localize the drug at the site of action; • (3) allow for release of the drug chemically or enzymatically; • (4) minimize host toxicity; • (5) biodegradable, biochemically inert, and non-immunogenic; • (6) easy prepare inexpensively • (7) Chemically and biochemically stable in its dosage form
- 11. • Most common (biologically labile) functional groups utilized in prodrug design are shown above.
- 12. • Esters are the most commonly employed prodrugs. • Numerous catalytic esterases are present in vivo to hydrolyze simple esters. Prodrug Active Form of Drug
- 13. • However, different species have differing amounts and types of esterases with different substrate specificities and different rates of hydrolysis. • This can make it difficult for pharmaceutical companies to generate accurate preclinical models in which to evaluate their candidate prodrug.
- 14. • One example is the monoethyl ester of enalaprilat, which is called enalapril. • Enalaprilate (upper left) was first discovered as an inhibitor of angiotensin converting enzyme (ACE) and used to treat hypertension. • Due to its high polarity, note two COOH’s, it was not orally bioavailable, and thus needed to be administered by injection. • The monomethyl ester, enalapril (upper right) is orally bioavailable.
- 15. • Another example is the anti-viral agent Oseltamavir (Tamiflu®) shown above • Notice that the oral bioavailability is improved by employing the ethyl ester of the carboxylic acid
- 16. Prodrugs • To minimize toxicity To Encourage Patient Acceptance • To eliminate formulation problems Improved absorption
- 17. Prodrugs • Increased water solubility •
- 18. Bioprecursors • Prontosil to sulfonamides
- 19. Bioprecursors • Activation of leflunomide to active drug
- 20. Bioprecursors • Sulfation activation Decarboxylation activation