Case by Case: Actioning Patient-Centered Strategies for Risk Assessment, Diagnosis, and Management of Amyloid-Related Imaging Abnormalities (ARIA)
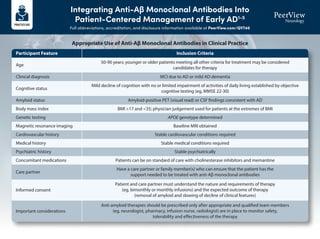
- 1. Integrating Anti-Aβ Monoclonal Antibodies Into Patient-Centered Management of Early AD1-5 Full abbreviations, accreditation, and disclosure information available at PeerView.com/QVT40 Participant Feature Age Clinical diagnosis Cognitive status Amyloid status Body mass index Genetic testing Magnetic resonance imaging Cardiovascular history Medical history Psychiatric history Concomitant medications Care partner Informed consent Important considerations 50-90 years; younger or older patients meeting all other criteria for treatment may be considered candidates for therapy MCI due to AD or mild AD dementia Mild decline of cognition with no or limited impairment of activities of daily living established by objective cognitive testing (eg, MMSE 22-30) Amyloid-positive PET (visual read) or CSF findings consistent with AD BMI >17 and <35; physician judgement used for patients at the extremes of BMI APOE genotype determined Baseline MRI obtained Stable cardiovascular conditions required Stable medical conditions required Stable psychiatrically Patients can be on standard of care with cholinesterase inhibitors and memantine Have a care partner or family member(s) who can ensure that the patient has the support needed to be treated with anti-Aβ monoclonal antibodies Patient and care partner must understand the nature and requirements of therapy (eg, bimonthly or monthly infusions) and the expected outcome of therapy (removal of amyloid and slowing of decline of clinical features) Anti-amyloid therapies should be prescribed only after appropriate and qualified team members (eg, neurologist, pharmacy, infusion nurse, radiologist) are in place to monitor safety, tolerability and effectiveness of the therapy Inclusion Criteria Appropriate Use of Anti-Aβ Monoclonal Antibodies in Clinical Practice
- 2. Integrating Anti-Aβ Monoclonal Antibodies Into Patient-Centered Management of Early AD1-5 Full abbreviations, accreditation, and disclosure information available at PeerView.com/QVT40 Participant Feature Cognitive impairment etiologies Baseline MRI findings Additional CNS injuries/findings Autoimmune disease/treatments Bleeding disorders Anticoagulants Other medical/psychiatric conditions Any medical, neurologic, or psychiatric condition that may be contributing to the cognitive impairment or any non-AD MCI or dementia • >4 microhemorrhages (defined as ≤10 mm at the greatest diameter) • A single macrohemorrhage (defined as >10 mm at the greatest diameter) • An area of superficial siderosis • Evidence of vasogenic edema • >2 lacunar infarcts or stroke involving a major vascular territory • Severe subcortical hyperintensities consistent with a Fazekas score of 3 • Evidence of ABRA • Evidence of CAA-ri • Any other major intracranial pathology that may cause cognitive impairment • MRI evidence of a non-AD dementia Recent history (within 12 mo) of stroke or TIA or any history of seizures Any history of immunologic disease (eg, lupus erythematosus, rheumatoid arthritis, Crohn’s disease) or systemic treatment with immunosuppressants, immunoglobulins, or monoclonal antibodies or their derivatives Patients with a bleeding disorder that is not under adequate control (including a platelet count <50,000 or INR >1.5 for participants who are not on anticoagulants) Patients on anticoagulants (coumadin, dabigatran, edoxaban, rivaroxaban, apixaban, betrixaban, or heparin) should not receive anti-Aβ monoclonal antibodies; tPA should not be administered to individuals on treatment with anti-Aβ monoclonal antibody • Mental illness (eg, psychosis) that interferes with comprehension of the requirements, potential benefit, and potential harms of treatment and are considered by the physician to render the patient unable to comply with management requirements • Major depression that will interfere with comprehension of the requirements, potential benefit, and potential harms of treatment; patients for whom disclosure of a positive biomarker may trigger suicidal ideation. Patients with less severe depression or whose depression resolves may be treatment candidates • Unstable medical conditions that may affect or be affected by treatment Exclusion Criteria Appropriate Use of Anti-Aβ Monoclonal Antibodies in Clinical Practice (Cont’d)
- 3. Integrating Anti-Aβ Monoclonal Antibodies Into Patient-Centered Management of Early AD1-5 Full abbreviations, accreditation, and disclosure information available at PeerView.com/QVT40 1. Cummings J et al. J Prev Alzheimers Dis. 2023;10:362-377. 2. Cummings J et al. J Prev Alzheimers Dis. 2022;2:221-230. 3. van Dyck CH et al. N Engl J Med. 2023;388:9-21. 4. Aduhelm (aducanumab) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/ label/2023/761178s007lbl.pdf. 5. Leqembi (lecanemab) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/761269s001lbl.pdf. 1. Clinician skilled in the assessment of cognition to identify individuals with MCI or mild dementia due to Alzheimer’s disease 2. MRI available for baseline assessment of cerebrovascular pathology and for monitoring of ARIA 3. Radiologists, neurologists, or other clinicians expert in the identification and interpretation of cerebrovascular lesions and ARIA 4. Amyloid PET or lumbar puncture capability to determine the amyloid status of treatment candidates 5. Radiologists, nuclear medicine specialists, neurologists, or other specialists skilled in the interpretation of amyloid imaging or neurologists, radiologists, or other clinicians skilled in the conduct of lumbar puncture 6. APOE genotyping resources 7. Genetic expertise to counsel patients on the implications of APOE genotyping 8. Expertise in communicating with patients and care partners regarding anticipated benefits, potential harm, and requirements for administration and monitoring while on anti-Aβ monoclonal antibodies 9. Infusion settings that can be made available to patients receiving therapy 10. Knowledgeable staff at infusion sites capable of recognizing and managing infusion reactions 11. Communication channels established between experts interpreting MRIs and clinicians treating patients with anti-Aβ monoclonal antibodies 12. Communication channels established between clinicians treating patients with anti-Aβ monoclonal antibodies and the patient and care partner 13. Availability of hospital resources including intensive care unit 14. Expertise in the management of seizures and status epilepticus patients with severe or serious ARIA 15. Protocol with standard operating procedures for management of serious and severe ARIA Resources Needed by a Clinician or Medical Center for the Safe and Effective Use of Anti-Aβ Monoclonal Antibodies in Clinical Practice
- 4. Detecting, Monitoring, and Treating Amyloid-Related Imaging Abnormalities (ARIA) Full abbreviations, accreditation, and disclosure information available at PeerView.com/QVT40 ARIA Type ARIA-E ARIA-H microhemorrhage ARIA-H superficial siderosis FLAIR hyperintensity confined to sulcus and/or cortex/subcortical white matter in one location <5 cm Mild Moderate Radiographic Severity Severe ≤4 new incident microhemorrhages One focal area of superficial siderosis FLAIR hyperintensity 5-10 cm, or more than one site of involvement, each measuring <10 cm 5-9 new incident microhemorrhages Two focal areas of superficial siderosis FLAIR hyperintensity measures >10 cm, often with significant subcortical white matter and/or sulcal involvement; ≥1 separate sites of involvement might be noted ≥10 new incident microhemorrhages >2 focal areas of superficial siderosis Classifying ARIA by Radiographic Severity1,2
- 5. Detecting, Monitoring, and Treating Amyloid-Related Imaging Abnormalities (ARIA) Full abbreviations, accreditation, and disclosure information available at PeerView.com/QVT40 3T scanner (recommended) 1.5T scanner (minimal) High field strength scanners have greater sensitivity but limited availability. The use of 1.5T scanner is endorsed as a minimum standard Slice thickness: ≤5 mm Thinner slices increase resolution, but decrease signal-to-noise ratio TE: ≥20 ms Longer TE increases sensitivity to detection 2D T2* GRE or SWI (for ARIA-H) To identify superficial siderosis and microhemorrhages (ARIA-H), T2* GRE and SWI are MRI sequences used to improve the detection and visualization of microhemorrhages T2-FLAIR (for ARIA-E) To monitor brain edema or sulcal effusion (ARIA-E) Diffusion-weighted imaging Recommended for differential diagnosis MRI Acquisition Protocols to Detect and Monitor ARIA3-5
- 6. Detecting, Monitoring, and Treating Amyloid-Related Imaging Abnormalities (ARIA) Full abbreviations, accreditation, and disclosure information available at PeerView.com/QVT40 Anti-Aβ antibodies require MRI assessment and monitoring before and during treatment • At baseline (up to one year prior to treatment initiation) • At recommended intervals (particularly during titration) • In response to symptoms suggesting possible ARIA • Important to obtain consistent serial imaging by using a standardized imaging protocol with the same field strength for every MRI assessment • After 12 months of treatment, decision about whether to continue to obtain safety MRIs should be guided by patient symptoms and prior MRI findings Recommended MRI Monitoring Schedules Aducanumab and lecanemab Aducanumab only Lecanemab only MRI within 1 year prior to initiation MRI if any symptoms suggestive of ARIA occur MRI prior to 5th dose MRI prior to 7th dose MRI prior to 9th dose MRI prior to 12th dose MRI prior to 14th dose T1 T2 T3 T4 T5 T6 T7 T8 T9 T12 T13 T14 Recommended MRI Monitoring Strategy to Detect ARIA in Patients Receiving Anti-Aβ Antibodies1-4,6-9
- 7. Detecting, Monitoring, and Treating Amyloid-Related Imaging Abnormalities (ARIA) Full abbreviations, accreditation, and disclosure information available at PeerView.com/QVT40 Headache Confusion and dizziness Neuropsychiatric symptoms Nausea Gait disturbance Visual disturbance/ blurred vision Seizure Less frequent Uncommon Symptom Severity Mild Discomfort noted; no disruption of daily activity Moderate Discomfort sufficient to reduce or affect normal daily activity Severe Incapacitating, with inability to perform normal daily activity Symptoms Consistent With ARIA That Should Trigger Out-of-Sequence MRI6-8
- 8. Detecting, Monitoring, and Treating Amyloid-Related Imaging Abnormalities (ARIA) Full abbreviations, accreditation, and disclosure information available at PeerView.com/QVT40 Baseline MRI has no exclusion factors MRI routine or conducted because of symptoms suggestive of ARIA ARIA-E or ARIA-H detected Symptomatic Asymptomatic Radiographically mild ARIA-E or mild ARIA-H Continue treatment with anti-Aβ antibody; monthly MRI Continue treatment; discontinue monthly MRI if ARIA-E resolves or ARIA-H stabilizes Resume treatment with anti-Aβ antibody Suspend treatment; clinical assessment; repeat MRI monthly Radiographically moderate/severe ARIA-E or moderate/severe ARIA-H MRI shows resolution of ARIA-E or stabilization of ARIA-H; symptoms resolve; patient wishes to continue Stop anti-Aβ antibody therapy for any of the following • Any macrohemorrhage • >1 area of superficial siderosis • >10 microhemorrhages since treatment initiation • >2 episodes of ARIA • Severe symptoms of ARIA • Patient requires treatment with an anticoagulant Severity of Changes Observed on MRI Radiographic ARIA-E: Mild Radiographic ARIA-E: Moderate Radiographic ARIA-E: Severe Radiographic ARIA-H: Mild Radiographic ARIA-H: Moderate Radiographic ARIA-H: Severe Symptom Description No Symptoms Mild Symptoms Moderate Symptoms Severe Symptoms Discontinue dosing Discontinue dosing Discontinue dosing Discontinue dosing Discontinue dosing Discontinue dosing Discontinue dosing Discontinue dosing Discontinue dosing Discontinue dosing Suspend dosing Suspend dosing Suspend dosing Suspend dosing Suspend dosing Suspend dosing Suspend dosing Suspend dosing Suspend dosing Suspend dosing Discontinue dosing Discontinue dosing Continue dosing Continue dosing ARIA Management Algorithm6-8
- 9. Detecting, Monitoring, and Treating Amyloid-Related Imaging Abnormalities (ARIA) Full abbreviations, accreditation, and disclosure information available at PeerView.com/QVT40 1. Aduhelm (aducanumab) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/761178s007lbl.pdf. 2. Leqembi (lecanemab) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/761269s000lbl.pdf. 3. Cogswell PM et al. AJNR Am J Neuroradiol. 2022;43:E19-E35. 4. Sperling RA et al. Alzheimers Dement. 2011;7:367-385. 5. Barakos J et al. J Prev Alzheimers Dis. 2022;9:211-220. 6. Cummings J et al. J Prev Alzheimers Dis. 2023;10:362-377. 7. Cummings J et al. J Prev Alzheimers Dis. 2022;2:221-230. 8. Cummings J et al. J Prev Alzheimers Dis. 2021;4:398-410. 9. van Dyck CH et al. N Engl J Med. 2023;388:9-21. • Referral of patient to emergency department for thorough assessment of suspected/known ARIA • Brain MRI without contrast enhancement if not already obtained (FLAIR, T2* GRE or SWI, and DWI sequences) • MRI review by a reader proficient in detection of ARIA (preferably with access to past MRIs for comparison) and rapid communication between MRI reader and clinicians responsible for patient’s treatment and AD care • Discontinuation of anti-amyloid therapy • Consultation by a neurologist • Admittance to hospital ward for close neurologic monitoring and tiered level of monitoring and management • Admit or transfer to a stroke care unit or neurological intensive care unit if warranted • When warranted, protocols for – Early initiation of treatment with IV methylprednisolone 1 g/d for 5 d – Conducting electroencephalography to detect epileptiform activity – Treatment with anticonvulsants for seizure management or prophylaxis if electroencephalography suggests they are indicated – Consideration of additional immunosuppressive treatment if not responding to methylprednisolone after 5 d of treatment – Plan transition to oral steroid treatment and taper as outpatient • Support and communicate with patient and family members/care partners throughout the event with informed patient-centered decision-making Triage Strategy for Severe Symptomatic Cases of ARIA7
- 10. Selected Images of Amyloid-Related Imaging Abnormalities (ARIA) and Its Mimics Full abbreviations, accreditation, and disclosure information available at PeerView.com/QVT40 Parenchymal Edema New T2-FLAIR hyperintense signal with mild local mass effect and sulcal effacement measuring <5 cm (mild ARIA-E) New multifocal, patchy T2-FLAIR hyperintense signal, each region measuring <5 cm (moderate ARIA-E); multiple ARIA-E yields a classification of moderate, as long as each region is <10 cm Extensive T2-FLAIR hyperintense signal throughout the right frontal and parietal lobes measuring >10 cm (severe ARIA-E); associated mass effect and sulcal effacement throughout much of the right hemisphere Baseline Baseline Baseline Post-treatment Post-treatment Post-treatment Mild Moderate Severe New sulcal T2-FLAIR hyperintense signal measuring <5 cm in transverse dimensions (mild ARIA-E) New T2-FLAIR sulcal effusion involving the right posterior temporal and parietal lobes measuring 5-10 cm (moderate ARIA-E) Extensive T2-FLAIR sulcal effusion involving the bilateral temporal and occipital lobes measuring ≥10 cm in extent (severe ARIA-E) Baseline Post-treatment Mild Sulcal Effusion Baseline Post-treatment Moderate Baseline Post-treatment Severe ARIA-E Examples (Detected With T2-FLAIR Sequence)1
- 11. Selected Images of Amyloid-Related Imaging Abnormalities (ARIA) and Its Mimics Full abbreviations, accreditation, and disclosure information available at PeerView.com/QVT40 Microhemorrhage Postdosing, ≥10 new microhemorrhages (severe ARIA-H) Baseline Post-treatment Severe Postdosing, 5 treatment-emergent microhemorrhages (moderate ARIA-H) Baseline Post-treatment Moderate Postdosing, few (<5) new peripheral left frontal microhemorrhages (mild ARIA-H) Baseline Post-treatment Mild Superficial Siderosis Postdosing, new right temporal superficial siderosis, which involves contiguous sulci when viewed over multiple slices (mild ARIA-H, siderosis); this patient also had two treatment-emergent microhemorrhages (mild ARIA-H, microhemorrhage) Two regions of treatment-emergent superficial siderosis in the right greater-than-left frontal lobes (moderate ARIA-H) Baseline Post-treatment Mild Baseline Post-treatment Moderate ARIA-H Examples (Detected With T2-GRE or SWI Sequence)1
- 12. Selected Images of Amyloid-Related Imaging Abnormalities (ARIA) and Its Mimics Full abbreviations, accreditation, and disclosure information available at PeerView.com/QVT40 Infarct (Early Subacute) FLAIR+ (hyperintensity) • In addition to FLAIR and GRE/SWI sequences, a trace DWI sequence should be included as routine protocol in ARIA monitoring examinations to help with the differential diagnosis of new signal abnormalities • For example, the DWI sequence plays an important role in helping to differentiate ARIA-E from potential cytotoxic edema caused by an incidental infarct • In classic cases of ARIA, the diffusion restriction will be absent, because intense diffusion restriction associated with an infarct is not a characteristic of ARIA • The DWI sequence helped to identify the underlying etiology of this patient’s radiographic findings • Diffusion restriction was identified, which indicated that it was not ARIA • However, the diffusion restriction was located in the sulci, which is not a typical pattern seen with acute or subacute infarcts • This patient was diagnosed with bacterial meningitis following confirmatory lab testing Infection (Bacterial Meningitis) FLAIR+ (vasogenic edema and sulcal effusions) GRE/SWI+ (microhemorrhages and superficial siderosis) DWI+ (with restricted diffusion in the sulci) DWI+ (with restricted diffusion) GRE/SWI+ (microhemorrhage) ARIA Mimics2
- 13. Selected Images of Amyloid-Related Imaging Abnormalities (ARIA) and Its Mimics Full abbreviations, accreditation, and disclosure information available at PeerView.com/QVT40 1. Cogswell PM et al. Am J Neuroradiol. 2022;43:E19-E35. 2. Images courtesy of Tammie L.S. Benzinger, MD, PhD. ARIA Mimics2 Brain Metastasis FLAIR+ (hyperintensity) • New signal abnormalities detected in ARIA monitoring examinations may require T1-weighted gadolinium-enhanced imaging to differentiate between ARIA and brain metastases • Contrast-enhanced imaging should be considered based on the patient’s medical history and/or any clinical findings that may suggest the possibility of a metastatic etiology • ARIA and PRES are usually indistinguishable based on MRI (eg, both will typically have FLAIR hyperintensities and no restricted diffusion) • Therefore, the clinical context is critical to differentiate between ARIA and PRES • This patient presented to the emergency department with confusion, headache, and very high blood pressure (which is a classic presentation for PRES) • They were treated for their hypertension, which resolved their clinical symptoms and their edema Posterior Reversible Encephalopathy Syndrome (PRES) FLAIR+ (hyperintensities) GRE/SWI negative DWI negative (no diffusion restriction) T1 postcontrast + enhancement GRE/SWI+ (microhemorrhage) T1 postcontrast
