Enabling higher process titers at a manufacturing facility
•
0 likes•149 views
10th— 11th - February 2015 – Radisson Blu, Heathrow Airport, Hayes, London, UK Annual BIOINNOVATION | Seamless, Integrated & Innovative BioManufacturing http://www.bioinnovation.gb.com/ Annual PIMS | 2015 |Pharmaceutical Innovation, Operational Excellence & Manufacturing Strategies Summit http://www.pims.gb.com/ 16th & 17th September 2015 | Boston Marriott Cambridge, MA | USA Annual ManuBIO | 2015 Flexible, Integrated & Innovative Biological Manufacturing Excellence USA http://www.manubio.com/ VIDEO WATCH
Report
Share
Report
Share
Download to read offline
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
The Future of Bioprocessing – What you need to know.

The Future of Bioprocessing – What you need to know.
Facilities for personlised medicine today and tomorrow - when redoing a batch...

Facilities for personlised medicine today and tomorrow - when redoing a batch...
Next Generation Bioprocessing adoption for mAbs – BioContinuum™ Platform Info...

Next Generation Bioprocessing adoption for mAbs – BioContinuum™ Platform Info...
Identifying Appropriate-Quality Pharmaceutical Raw Materials in an Evolving R...

Identifying Appropriate-Quality Pharmaceutical Raw Materials in an Evolving R...
Detailed Professional Resume for Chris Harding: Chemical Engineer/Biological ...

Detailed Professional Resume for Chris Harding: Chemical Engineer/Biological ...
From preclinical to commercial: The evolution of ADC manufacturing expertise ...

From preclinical to commercial: The evolution of ADC manufacturing expertise ...
Challenges using Multiple Single-use Systems: Functionality versus Extractabl...

Challenges using Multiple Single-use Systems: Functionality versus Extractabl...
Similar to Enabling higher process titers at a manufacturing facility
Similar to Enabling higher process titers at a manufacturing facility (20)
The value of strategic partnerships to build technological leadership - Chris...

The value of strategic partnerships to build technological leadership - Chris...
Accenture Betting On Science Study Overview V2.0 12458298 Mr

Accenture Betting On Science Study Overview V2.0 12458298 Mr
Retrofitting a new process to an existing facility

Retrofitting a new process to an existing facility
How to Optimize Clean-in-Place (CIP) Processes in Food and Beverage Operations

How to Optimize Clean-in-Place (CIP) Processes in Food and Beverage Operations
More from GBX Summits
More from GBX Summits (16)
Analyzing the relationship between titer and processing time

Analyzing the relationship between titer and processing time
The Changing Pharma R&D Model, GDDIS Leaders Summit Ted Torphy CSO, Head, Ex...

The Changing Pharma R&D Model, GDDIS Leaders Summit Ted Torphy CSO, Head, Ex...
Qumas Collaboration to Innovation Quality QMS PIMS 2014

Qumas Collaboration to Innovation Quality QMS PIMS 2014
Dr. Elke Prohaska & Regina Römling BioInnovation Leader Summit Tosoh

Dr. Elke Prohaska & Regina Römling BioInnovation Leader Summit Tosoh
BioInnovation Leader Summit Transforming Process Efficiency

BioInnovation Leader Summit Transforming Process Efficiency
Recently uploaded
Saudi Arabia [ Abortion pills) Jeddah/riaydh/dammam/++918133066128☎️] cytotec tablets uses abortion pills 💊💊 How effective is the abortion pill? 💊💊 +918133066128) "Abortion pills in Jeddah" how to get cytotec tablets in Riyadh " Abortion pills in dammam*💊💊 The abortion pill is very effective. If you’re taking mifepristone and misoprostol, it depends on how far along the pregnancy is, and how many doses of medicine you take:💊💊 +918133066128) how to buy cytotec pills
At 8 weeks pregnant or less, it works about 94-98% of the time. +918133066128[ 💊💊💊 At 8-9 weeks pregnant, it works about 94-96% of the time. +918133066128) At 9-10 weeks pregnant, it works about 91-93% of the time. +918133066128)💊💊 If you take an extra dose of misoprostol, it works about 99% of the time. At 10-11 weeks pregnant, it works about 87% of the time. +918133066128) If you take an extra dose of misoprostol, it works about 98% of the time. In general, taking both mifepristone and+918133066128 misoprostol works a bit better than taking misoprostol only. +918133066128 Taking misoprostol alone works to end the+918133066128 pregnancy about 85-95% of the time — depending on how far along the+918133066128 pregnancy is and how you take the medicine. +918133066128 The abortion pill usually works, but if it doesn’t, you can take more medicine or have an in-clinic abortion. +918133066128 When can I take the abortion pill?+918133066128 In general, you can have a medication abortion up to 77 days (11 weeks)+918133066128 after the first day of your last period. If it’s been 78 days or more since the first day of your last+918133066128 period, you can have an in-clinic abortion to end your pregnancy.+918133066128
Why do people choose the abortion pill? Which kind of abortion you choose all depends on your personal+918133066128 preference and situation. With+918133066128 medication+918133066128 abortion, some people like that you don’t need to have a procedure in a doctor’s office. You can have your medication abortion on your own+918133066128 schedule, at home or in another comfortable place that you choose.+918133066128 You get to decide who you want to be with during your abortion, or you can go it alone. Because+918133066128 medication abortion is similar to a miscarriage, many people feel like it’s more “natural” and less invasive. And some+918133066128 people may not have an in-clinic abortion provider close by, so abortion pills are more available to+918133066128 them. +918133066128 Your doctor, nurse, or health center staff can help you decide which kind of abortion is best for you. +918133066128 More questions from patients: Saudi Arabia+918133066128 CYTOTEC Misoprostol Tablets. Misoprostol is a medication that can prevent stomach ulcers if you also take NSAID medications. It reduces the amount of acid in your stomach, which protects your stomach lining. The brand name of this medication is Cytotec®.+918133066128) Unwanted Kit Mifty kit IN Salmiya (+918133066128) Abortion pills IN Salmiyah Cytotec pills

Mifty kit IN Salmiya (+918133066128) Abortion pills IN Salmiyah Cytotec pillsAbortion pills in Kuwait Cytotec pills in Kuwait
Saudi Arabia [ Abortion pills) Jeddah/riaydh/dammam/++918133066128☎️] cytotec tablets uses abortion pills 💊💊 How effective is the abortion pill? 💊💊 +918133066128) "Abortion pills in Jeddah" how to get cytotec tablets in Riyadh " Abortion pills in dammam*💊💊 The abortion pill is very effective. If you’re taking mifepristone and misoprostol, it depends on how far along the pregnancy is, and how many doses of medicine you take:💊💊 +918133066128) how to buy cytotec pills
At 8 weeks pregnant or less, it works about 94-98% of the time. +918133066128[ 💊💊💊 At 8-9 weeks pregnant, it works about 94-96% of the time. +918133066128) At 9-10 weeks pregnant, it works about 91-93% of the time. +918133066128)💊💊 If you take an extra dose of misoprostol, it works about 99% of the time. At 10-11 weeks pregnant, it works about 87% of the time. +918133066128) If you take an extra dose of misoprostol, it works about 98% of the time. In general, taking both mifepristone and+918133066128 misoprostol works a bit better than taking misoprostol only. +918133066128 Taking misoprostol alone works to end the+918133066128 pregnancy about 85-95% of the time — depending on how far along the+918133066128 pregnancy is and how you take the medicine. +918133066128 The abortion pill usually works, but if it doesn’t, you can take more medicine or have an in-clinic abortion. +918133066128 When can I take the abortion pill?+918133066128 In general, you can have a medication abortion up to 77 days (11 weeks)+918133066128 after the first day of your last period. If it’s been 78 days or more since the first day of your last+918133066128 period, you can have an in-clinic abortion to end your pregnancy.+918133066128
Why do people choose the abortion pill? Which kind of abortion you choose all depends on your personal+918133066128 preference and situation. With+918133066128 medication+918133066128 abortion, some people like that you don’t need to have a procedure in a doctor’s office. You can have your medication abortion on your own+918133066128 schedule, at home or in another comfortable place that you choose.+918133066128 You get to decide who you want to be with during your abortion, or you can go it alone. Because+918133066128 medication abortion is similar to a miscarriage, many people feel like it’s more “natural” and less invasive. And some+918133066128 people may not have an in-clinic abortion provider close by, so abortion pills are more available to+918133066128 them. +918133066128 Your doctor, nurse, or health center staff can help you decide which kind of abortion is best for you. +918133066128 More questions from patients: Saudi Arabia+918133066128 CYTOTEC Misoprostol Tablets. Misoprostol is a medication that can prevent stomach ulcers if you also take NSAID medications. It reduces the amount of acid in your stomach, which protects your stomach lining. The brand name of this medication is Cytotec®.+918133066128) Unwanted Kit is a combination of two medicines, ounwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE AbudhabiAbortion pills in Kuwait Cytotec pills in Kuwait
VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS Live
Booking Contact Details :-
WhatsApp Chat :- [+91-9999965857 ]
The Best Call Girls Delhi At Your Service
Russian Call Girls Delhi Doing anything intimate with can be a wonderful way to unwind from life's stresses, while having some fun. These girls specialize in providing sexual pleasure that will satisfy your fetishes; from tease and seduce their clients to keeping it all confidential - these services are also available both install and outcall, making them great additions for parties or business events alike. Their expert sex skills include deep penetration, oral sex, cum eating and cum eating - always respecting your wishes as part of the experience
(07-May-2024(PSS)VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS...

VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS...Call Girls In Delhi Whatsup 9873940964 Enjoy Unlimited Pleasure
Recently uploaded (20)
KYC-Verified Accounts: Helping Companies Handle Challenging Regulatory Enviro...

KYC-Verified Accounts: Helping Companies Handle Challenging Regulatory Enviro...
The Coffee Bean & Tea Leaf(CBTL), Business strategy case study

The Coffee Bean & Tea Leaf(CBTL), Business strategy case study
Mifty kit IN Salmiya (+918133066128) Abortion pills IN Salmiyah Cytotec pills

Mifty kit IN Salmiya (+918133066128) Abortion pills IN Salmiyah Cytotec pills
The Path to Product Excellence: Avoiding Common Pitfalls and Enhancing Commun...

The Path to Product Excellence: Avoiding Common Pitfalls and Enhancing Commun...
MONA 98765-12871 CALL GIRLS IN LUDHIANA LUDHIANA CALL GIRL

MONA 98765-12871 CALL GIRLS IN LUDHIANA LUDHIANA CALL GIRL
unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi
VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS...

VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS...
Russian Call Girls In Gurgaon ❤️8448577510 ⊹Best Escorts Service In 24/7 Delh...

Russian Call Girls In Gurgaon ❤️8448577510 ⊹Best Escorts Service In 24/7 Delh...
Call Girls Pune Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Pune Just Call 9907093804 Top Class Call Girl Service Available
Call Girls Electronic City Just Call 👗 7737669865 👗 Top Class Call Girl Servi...

Call Girls Electronic City Just Call 👗 7737669865 👗 Top Class Call Girl Servi...
Enhancing and Restoring Safety & Quality Cultures - Dave Litwiller - May 2024...

Enhancing and Restoring Safety & Quality Cultures - Dave Litwiller - May 2024...
Ensure the security of your HCL environment by applying the Zero Trust princi...

Ensure the security of your HCL environment by applying the Zero Trust princi...
VIP Call Girls In Saharaganj ( Lucknow ) 🔝 8923113531 🔝 Cash Payment (COD) 👒

VIP Call Girls In Saharaganj ( Lucknow ) 🔝 8923113531 🔝 Cash Payment (COD) 👒
Call Girls In DLf Gurgaon ➥99902@11544 ( Best price)100% Genuine Escort In 24...

Call Girls In DLf Gurgaon ➥99902@11544 ( Best price)100% Genuine Escort In 24...
B.COM Unit – 4 ( CORPORATE SOCIAL RESPONSIBILITY ( CSR ).pptx

B.COM Unit – 4 ( CORPORATE SOCIAL RESPONSIBILITY ( CSR ).pptx
Enabling higher process titers at a manufacturing facility
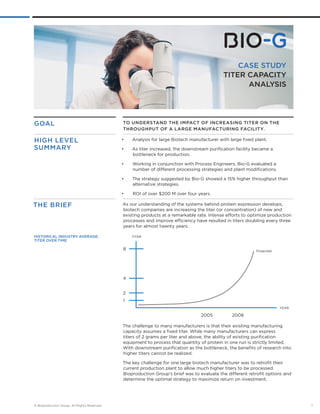
- 1. CASE STUDY TITER CAPACITY ANALYSIS To understand the impact of increasin g titer on the throu ghput of a lar ge manufacturin g facility . • Analysis for large Biotech manufacturer with large fixed plant. • As titer increased, the downstream purification facility became a bottleneck for production. • Working in conjunction with Process Engineers, Bio-G evaluated a number of different processing strategies and plant modifications. • The strategy suggested by Bio-G showed a 15% higher throughput than alternative strategies. • ROI of over $200 M over four years. As our understanding of the systems behind protein expression develops, biotech companies are increasing the titer (or concentration) of new and existing products at a remarkable rate. Intense efforts to optimize production processes and improve efficiency have resulted in titers doubling every three years for almost twenty years. 8 4 2 1 2005 Projected 2008 YEAR TITER GOAL high level summary THE BRIEF Historical Industry Average: Titer over Time The challenge to many manufacturers is that their existing manufacturing capacity assumes a fixed titer. While many manufacturers can express titers of 2 grams per liter and above, the ability of existing purification equipment to process that quantity of protein in one run is strictly limited. With downstream purification as the bottleneck, the benefits of research into higher titers cannot be realized. The key challenge for one large biotech manufacturer was to retrofit their current production plant to allow much higher titers to be processed. Bioproduction Group’s brief was to evaluate the different retrofit options and determine the optimal strategy to maximize return on investment. © Bioproduction Group. All Rights Reserved. 1
- 2. How We Did It “Establishing a credible estimate based on physical plant capability is the key to understanding value.” David Zhang, Principal Results Bio -G Results: 10-15% impro vement over proposed scenario MORE INFORMATION BIOPRODUCTION GROUP CONTACT@BIO-G.COM www.bio-g.com Bioproduction Group’s methodology was to firstly understand the impact of high titer on plant throughput. “The key question to ask is not about cost, but about runs per week,” comments Principal David Zhang. “Establishing a credible estimate based on physical plant capability is the key to understanding value.” Working with engineers in process development, Bio-G established a detailed simulation baseline that included all downstream main operations, buffer preparation steps, cleaning activities and support steps. The simulation was based directly from historical plant data and validated using process engineers on the shop floor. From this detailed simulation model, Bio-G built a number of scenarios representing the different ways in which the plant could process the additional protein product. One Engineer commented: “We needed someone who’d get down and dirty. Bio-G worked to deliver hard answers to hard questions.” Bio-G’s recommendations were to pursue a processing strategy based on the effect of variability on one key bottleneck step in downstream operations. This single processing step was shown to dominate the overall processing time and value of the scenario. Based on this recommendation, the plant was able to increase their overall plant throughput by 10-15% compared to the next best high titer scenario. TIME (hours) 6000 5000 4000 3000 2000 1000 20 40 60 80 100 120 NUMBER OF OUTPUT BATCHES The strategy allowed the company to minimize additional investment in infrastructure, while nearly doubling effective plant throughput. The ROI of the scenario was over $200 M in a period of four years. © Bioproduction Group. All Rights Reserved. 2
